
Generally, substances are identified (qualitatively) by the order in which they elute from the column and by the retention time of the analyte in the column. A detector is used to monitor the time at which each component reaches the outlet and ultimately the amount of that component can be determined. Since each type of molecule has a different rate of progression, the various components of the analyte mixture are separated as they progress along the column and reach the end of the column at different times (retention time). As the carrier gas transports the analyte molecules through the column, there is adsorption of the analyte molecules either onto the column walls or onto packing materials (stationary phase) in the column to give separation. In a GC analysis, a known volume of gaseous or liquid analyte is injected through a rubber disk and into a hot, temperature controlled, port attached to the column.
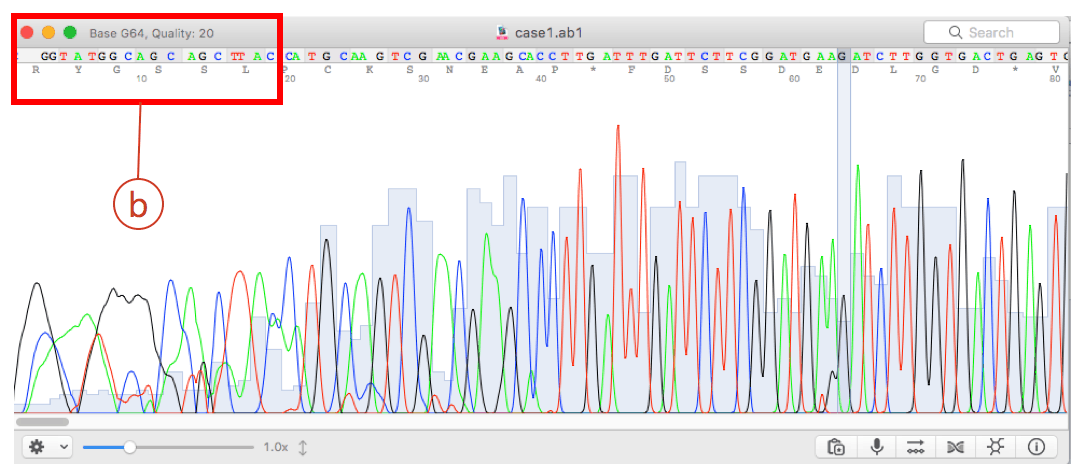
Other parameters that can be used to alter the order or time of retention are the carrier gas flow rate, column length and the temperature. The function of the stationary phase in the column is to separate different components, causing each one to exit the column at a different time.
As the chemicals exit the end of the column, they are detected and identified electronically. A gas chromatograph is made up of a narrow flow-through tube, known as the column, through which the sample passes in a gas stream (the carrier gas) at different rates depending on their various chemical and physical properties and their interaction with a specific column lining or filling, called the 'stationary phase'. GC analysis Ī gas chromatograph is a chemical analysis instrument for separating chemicals in a complex sample. The popularity of gas chromatography quickly rose after the development of the flame ionization detector. English chemists Archer Martin and Richard Synge received a Nobel Prize in 1952 for the invention of partition chromatography in the 1940s, laying the foundations of gas chromatography. German physical chemist Erika Cremer in 1947 together with Austrian graduate student Fritz Prior developed the theoretical foundations of GC and built the first liquid-gas chromatograph, but her work was deemed irrelevant and was ignored for a long time. Ĭhromatography dates to 1903 in the work of the Russian scientist, Mikhail Semenovich Tswett, who separated plant pigments via liquid column chromatography. The glass or metal column through which the gas phase passes is located in an oven where the temperature of the gas can be controlled and the eluent coming off the column is monitored by a computerized detector. The surface of the solid particles may also act as the stationary phase in some columns. The stationary phase is a microscopic layer of viscous liquid on a surface of solid particles on an inert solid support inside a piece of glass or metal tubing called a column. The mobile phase is usually an inert gas or an unreactive gas such as helium, argon, nitrogen or hydrogen. Gas chromatography is the process of separating compounds in a mixture by injecting a gaseous or liquid sample into a mobile phase, typically called the carrier gas, and passing the gas through a stationary phase. These alternative names, as well as their respective abbreviations, are frequently used in scientific literature. Gas chromatography is also sometimes known as vapor-phase chromatography (VPC), or gas–liquid partition chromatography (GLPC). In preparative chromatography, GC can be used to prepare pure compounds from a mixture. Typical uses of GC include testing the purity of a particular substance, or separating the different components of a mixture. Gas chromatography ( GC) is a common type of chromatography used in analytical chemistry for separating and analyzing compounds that can be vaporized without decomposition. msc (Management Saved Console).A gas chromatograph with a headspace sampler
